In this video, we will focus on paper chromatography.
Chromatography and Chromatogram
Chromatography is a physical method used to separate two or more components that dissolve in the same solvent. One type of chromatography is paper chromatography. Another important type is gas chromatography. We will focus on paper chromatography in this video.
We can use chromatography to separate and identify components in a sample. We can also use chromatography to determine purity of a substance. A pure substance gives only one spot on a chromatogram.
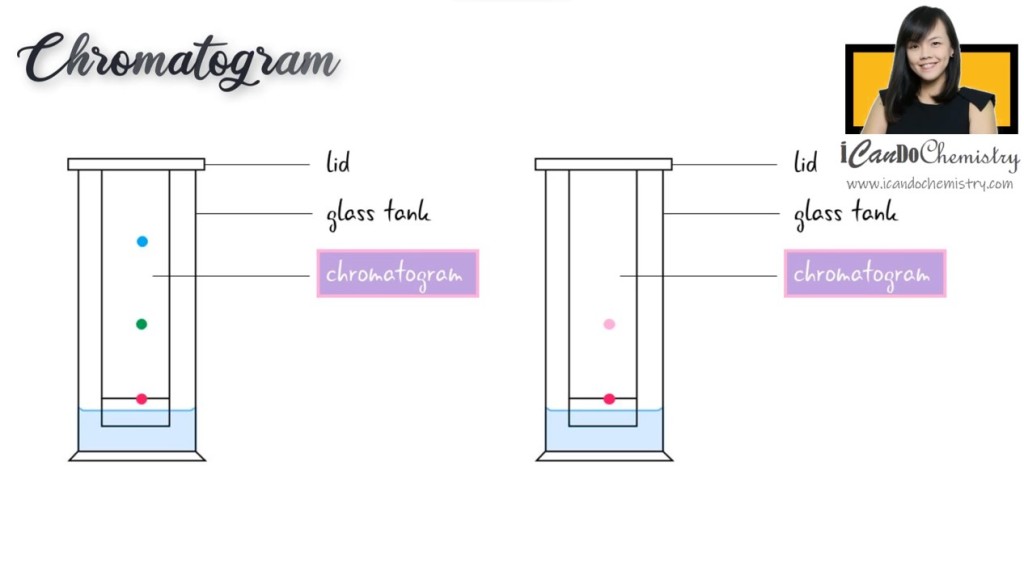
So, what is chromatogram? Chromatogram is the result of chromatography, showing us the separated components of a sample.
Setting up paper chromatography
We draw a line at the bottom of the chromatography paper, about 1 cm from the bottom of this strip will do. Do note, it is important that we use pencil instead of pen for this line because we do not want the ink from the pen to dissolve in the solvent and interfere with the result.
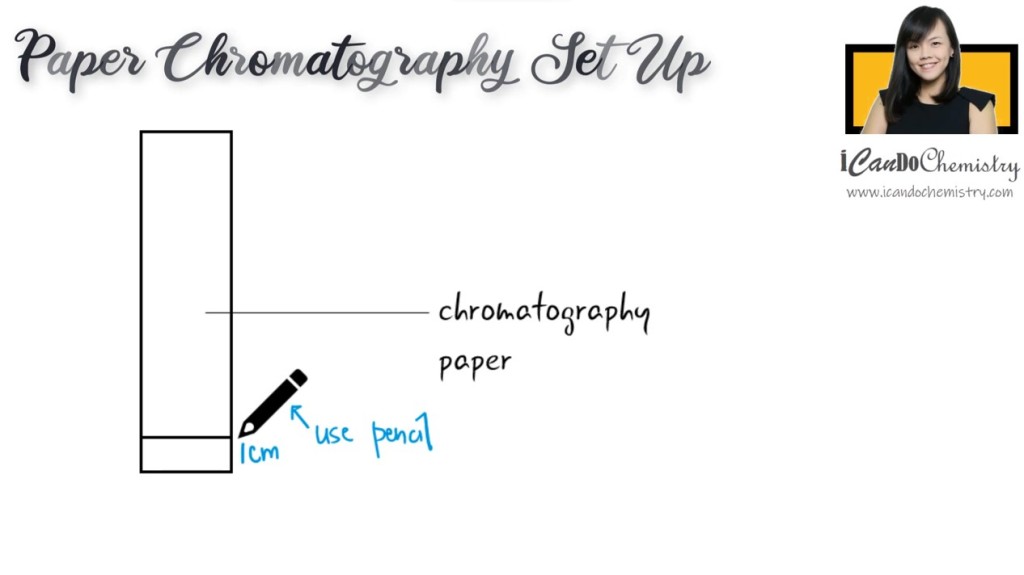
Then, we put a small drop of the sample on the pencil line.
Once the sample has dried, we dip the chromatography paper into a suitable solvent. There are different solvents available, such as ethanol. The solvent will then get soaked up by the paper, and it will travel up the paper and dissolve the sample. The sample will travel up the chromatography paper together with the solvent. Different components in the sample will move at different speeds, and so become separated.
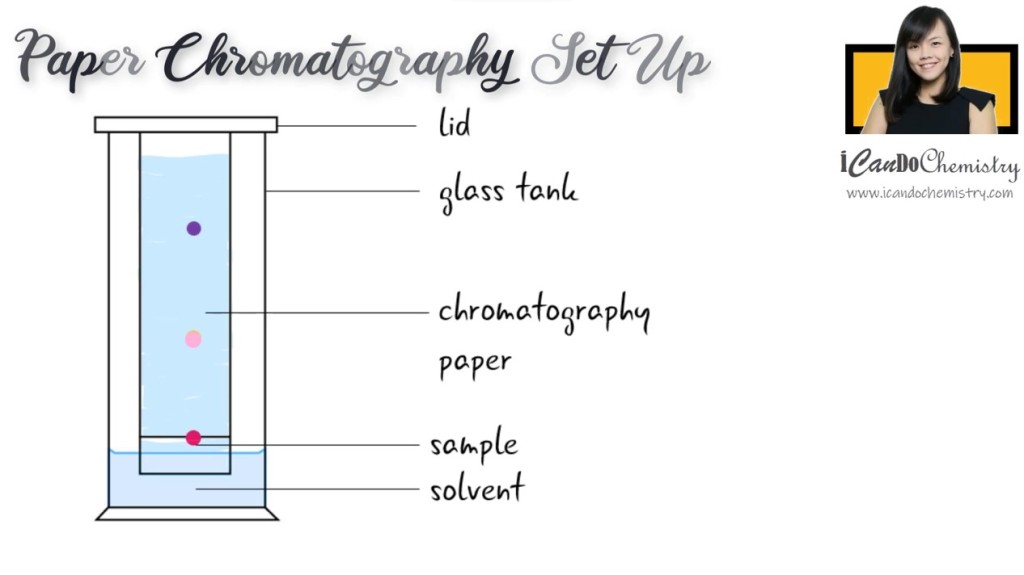
We will leave the apparatus to stand. Do not shake or move the set up until the solvent has travelled up almost the end of the chromatography paper.
Interpretation of results
At the end of the experiment, you would achieve a chromatography paper with the separated components. We call this the chromatogram.
The sample that we used contains a mixture of components. On the other hand, if the sample that we use is a pure substance, it should only leave one spot on the chromatogram.
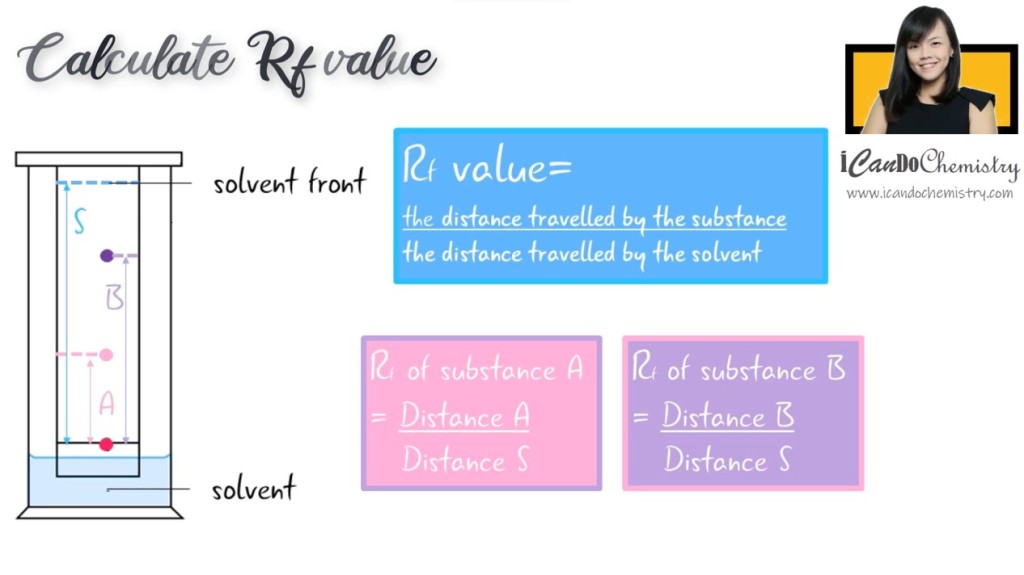
So what are these components present in the sample? We can determine that by calculating the Rf value, which is, the distance travelled by the substance divided by the distance travelled by the solvent. Different substances in a sample will have its own Rf values. We can then identify the substance by comparing its Rf value with Rf value of a known substance.
Questions
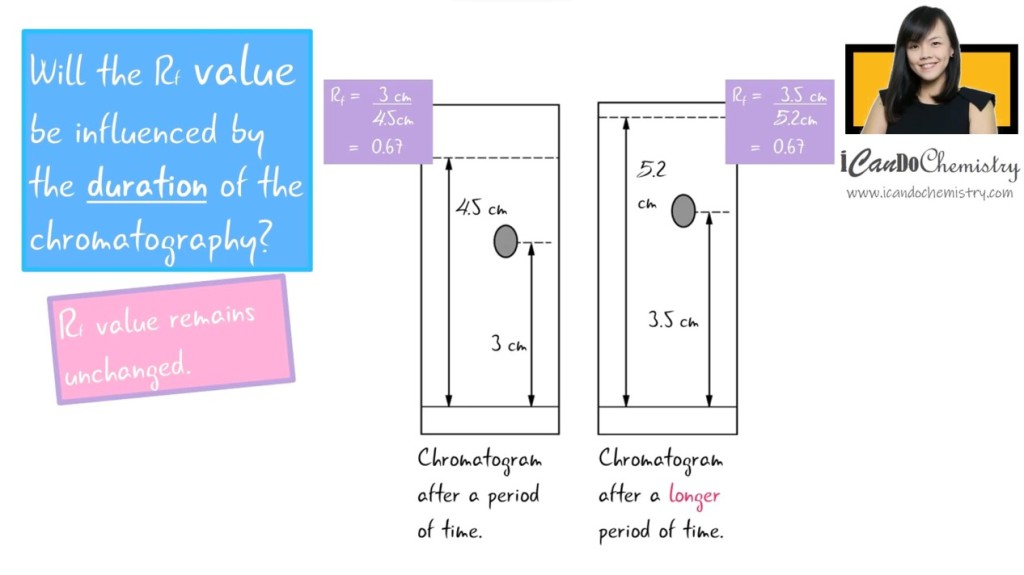
1. Will the Rf value be influenced by the duration of the chromatography? No. If we give it more time for the solvent to travel up the chromatography paper, the distance travelled by the component will increase too. Hence, Rf value remains the same. This is so if the conditions of the chromatography are the same – the same solvent is used and the temperature of the room is the same.
Like the Chemistry learning video and share with your friends if you find it useful. Don’t forget to subscribe to the channel for more Chemistry learning videos. Have fun learning Chemistry 😊
